Publications
2025 • 2024 • 2023 • 2022 • 2021 • 2020 • 2019 • 2018 • 2017 • 2016 • 2015 • 2014
Representative Covers from the Group


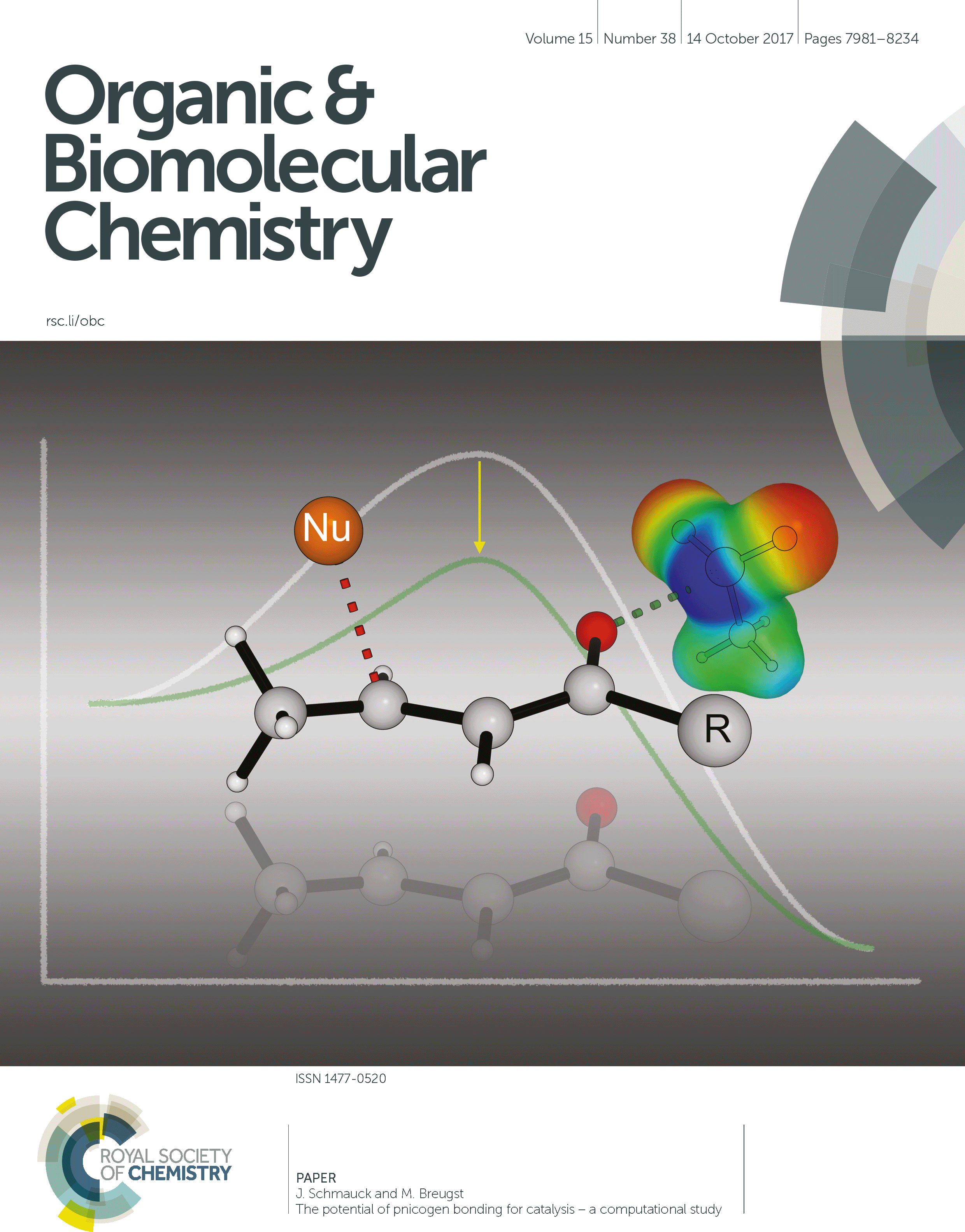
2025
82. Asymmetric Synthesis of C–N Axially Chiral Carbazoles via Axial-to-Axial Chirality Transfer
S. Myllek, P. Lencer, M. K. T. Klischan, B. Henssen, P. Neudecker, M. Breugst,* J. Pietruszka,* Org. Biomol. Chem., 2025, 23, 4888-4892.
DOI: 10.1039/D5OB00587F
S. Myllek, P. Lencer, M. K. T. Klischan, B. Henssen, P. Neudecker, M. Breugst,* J. Pietruszka,* Org. Biomol. Chem., 2025, 23, 4888-4892.
DOI: 10.1039/D5OB00587F

81. Trendberichte Organische Chemie 2025
M. Breugst,* J. Andexer, L. Barra, S. B. Beil, S. Bierbach, I. Burkhardt, O. Dumele, M. Ernst, J. Frommer, U. Gellrich, P. Germer, M. Giese, P. Huy, J. Klepp, K. Körber, M. Kordes, C. A. Kuttruff, T. Lindel, F. Pfrengle, B. Pieber, J. Pietruszka, N. Schaschke, M. O. Senge, N. Stadler, G. Storch, J. F. Teichert, S. R. Waldvogel, T. Werner, C. Winter, Nachr. Chem., 2025, 73, 40–70.
DOI: 10.1002/nadc.20254147860
M. Breugst,* J. Andexer, L. Barra, S. B. Beil, S. Bierbach, I. Burkhardt, O. Dumele, M. Ernst, J. Frommer, U. Gellrich, P. Germer, M. Giese, P. Huy, J. Klepp, K. Körber, M. Kordes, C. A. Kuttruff, T. Lindel, F. Pfrengle, B. Pieber, J. Pietruszka, N. Schaschke, M. O. Senge, N. Stadler, G. Storch, J. F. Teichert, S. R. Waldvogel, T. Werner, C. Winter, Nachr. Chem., 2025, 73, 40–70.
DOI: 10.1002/nadc.20254147860
80. A Unique Intramolecular Redox Reaction by Unidirectional Migration of Three Hydrogen Atoms: Theoretical Elucidation of the 3H-Transfer Sequence
T. Arndt, M. Schäfer, D. Kuck, M. Breugst,* Chem. Eur. J., 2025, 31, e202404638.
DOI: 10.1002/chem.202404638
T. Arndt, M. Schäfer, D. Kuck, M. Breugst,* Chem. Eur. J., 2025, 31, e202404638.
DOI: 10.1002/chem.202404638

79. Site-Selective Copper(I) Catalyzed Hydrogenation of Amides
D.-I. Tzaras, T. Jacquemin, M. Gorai, T. Arndt, B.M. Zimmermann, M. Breugst, J.F. Teichert, J. Am. Chem. Soc., 2025, 147, 1867–1874.
DOI: 10.1021/jacs.4c14174
also published as a preprint: ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-rz1j4.
Highlighted in Chemistry Views and in Synfacts 2025, 21, 270.
D.-I. Tzaras, T. Jacquemin, M. Gorai, T. Arndt, B.M. Zimmermann, M. Breugst, J.F. Teichert, J. Am. Chem. Soc., 2025, 147, 1867–1874.
DOI: 10.1021/jacs.4c14174
also published as a preprint: ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-rz1j4.
Highlighted in Chemistry Views and in Synfacts 2025, 21, 270.

2024
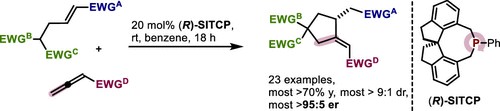
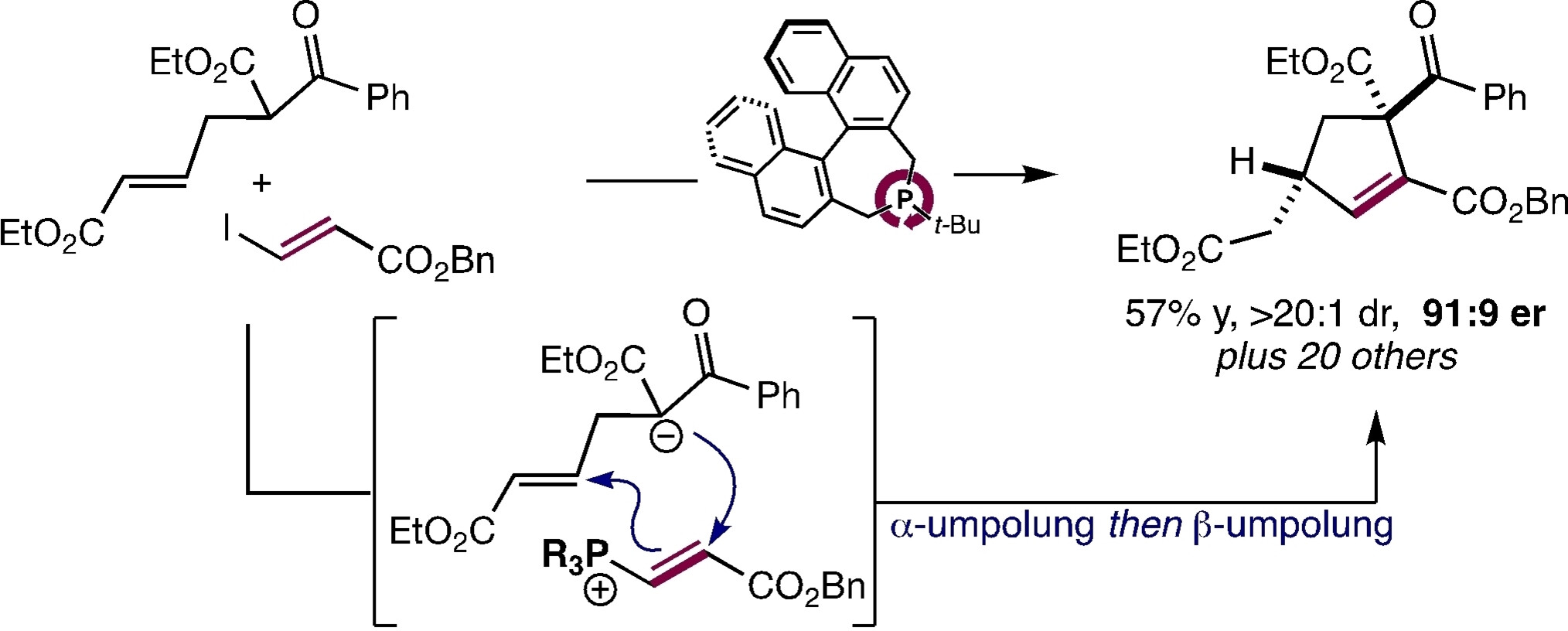
78. Enantioselective Synthesis of Cyclopentanes by Phosphine-Catalyzed β,γ-Annulation of Allenoates
C. Zhang, J. T. Maddigan-Wyatt, X. Nguyen, A. Seitz, M. Breugst, D. W. Lupton, Org. Lett., 2024, 26, 7800–7804.
DOI: 10.1021/acs.orglett.4c02371
C. Zhang, J. T. Maddigan-Wyatt, X. Nguyen, A. Seitz, M. Breugst, D. W. Lupton, Org. Lett., 2024, 26, 7800–7804.
DOI: 10.1021/acs.orglett.4c02371
77. Reactivity of Phosphaethynolate Anion with Stabilized Carbocations: Mechanistic Studies and Synthetic Applications
T.-H.-V. Nguyen, S. Chelli, S. Mallet-Ladeira, M. Breugst,* S. Lakhdar,* Chem. Sci., 2024, 15, 14406–14414.
DOI: 10.1039/D4SC03518F
T.-H.-V. Nguyen, S. Chelli, S. Mallet-Ladeira, M. Breugst,* S. Lakhdar,* Chem. Sci., 2024, 15, 14406–14414.
DOI: 10.1039/D4SC03518F

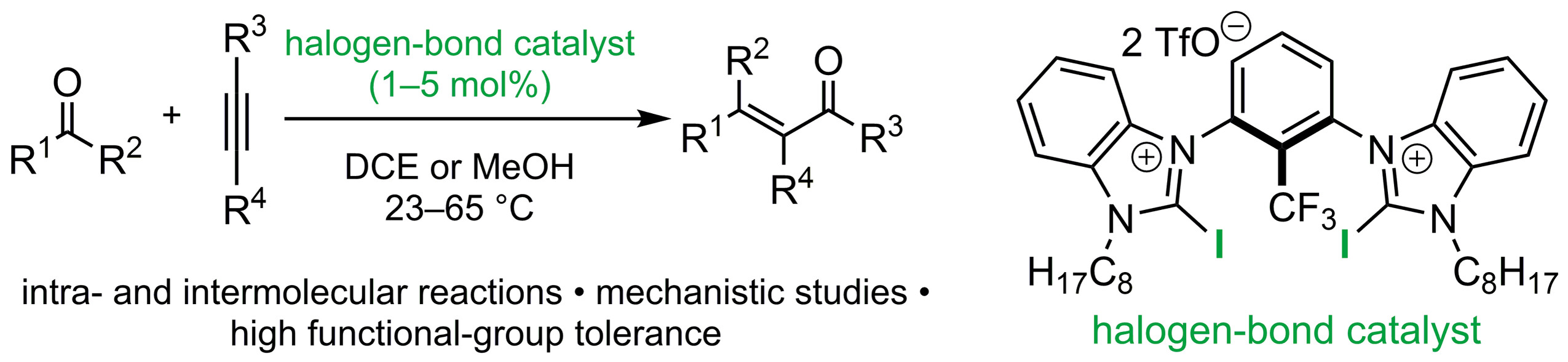
76. Carbonyl-Alkyne Metathesis Reactions Catalyzed by Organic Halogen-Bond Donors
T. Arndt, H. Ghazi Zahedi, M. Breugst,* Eur. J. Org. Chem., 2024, 27, e202400595.
DOI: 10.1002/ejoc.202400595
Invited contribution to the special collection Physical Organic Chemistry.
T. Arndt, H. Ghazi Zahedi, M. Breugst,* Eur. J. Org. Chem., 2024, 27, e202400595.
DOI: 10.1002/ejoc.202400595
Invited contribution to the special collection Physical Organic Chemistry.
75. Enantioselective synthesis of cyclopentenes by (2 + 3) cycloaddition via a 2-carbon phosphonium.
A. Seitz, J. T. Maddigan-Wyatt, J. Cao, M. Breugst, D. W. Lupton Angew. Chem. Int. Ed., 2024, 63, e202408397; Angew. Chem., 2024, 136, e202408397.
DOI: 10.1002/anie.2024083978
also published as a preprint: ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-9fk4c.
A. Seitz, J. T. Maddigan-Wyatt, J. Cao, M. Breugst, D. W. Lupton Angew. Chem. Int. Ed., 2024, 63, e202408397; Angew. Chem., 2024, 136, e202408397.
DOI: 10.1002/anie.2024083978
also published as a preprint: ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-9fk4c.
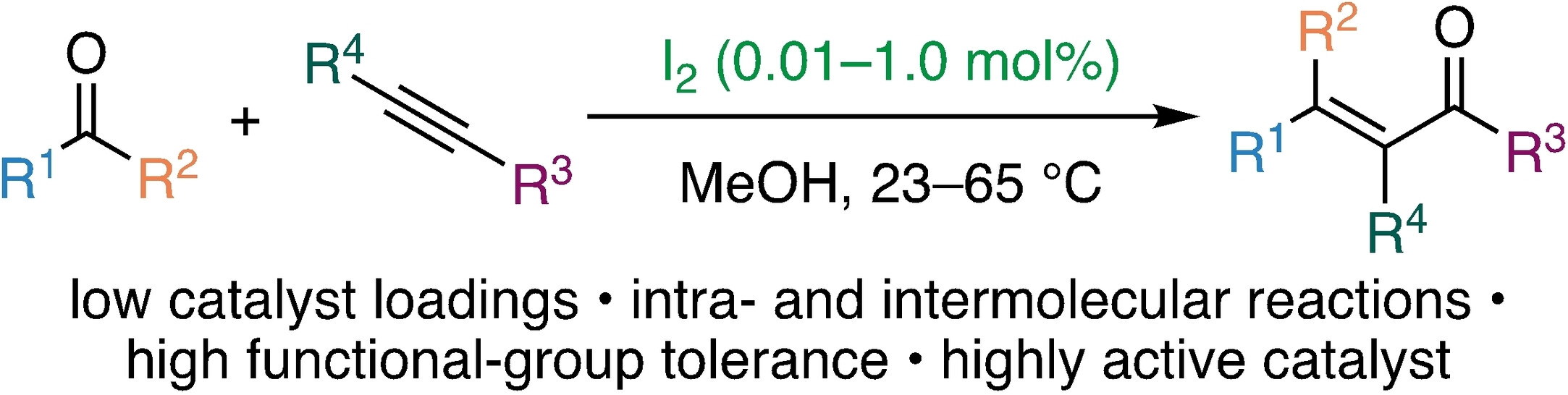
74. Iodine-Catalyzed Carbonyl-Alkyne Metathesis Reactions
T. Arndt, M. Breugst,* Chem. Eur. J., 2024, 30, e202402424.
DOI: 10.1002/chem.202402424
also published as a preprint: ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-4l3t8.
T. Arndt, M. Breugst,* Chem. Eur. J., 2024, 30, e202402424.
DOI: 10.1002/chem.202402424
also published as a preprint: ChemRxiv, 2024, DOI: 10.26434/chemrxiv-2024-4l3t8.
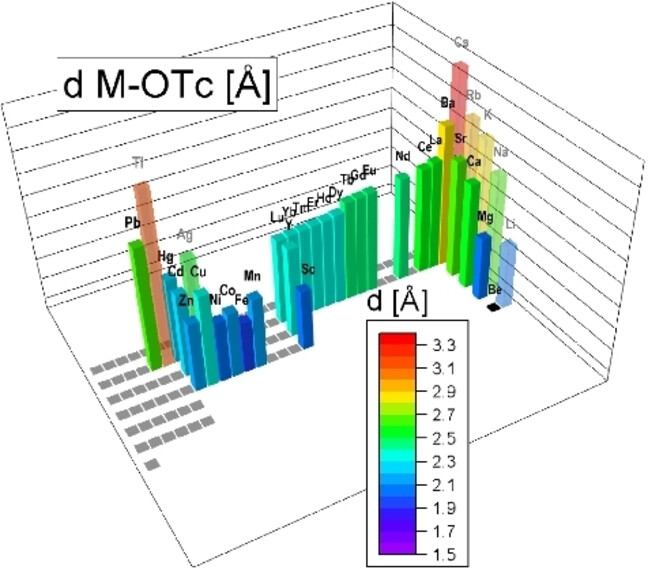
73. Pertechnetates – a structural study across the periodic table
E. Strub,* D. Grödler,* D. Zaratti, C. Yong, L. Dünnebier, S. Bazhenova, M. Roca Jungfer, M. Breugst,* M. Zegke* Chem. Eur. J., 2024, 30, e202400131.
DOI: 10.1002/chem.202400131
E. Strub,* D. Grödler,* D. Zaratti, C. Yong, L. Dünnebier, S. Bazhenova, M. Roca Jungfer, M. Breugst,* M. Zegke* Chem. Eur. J., 2024, 30, e202400131.
DOI: 10.1002/chem.202400131
72. Two distinct protocols for the synthesis of unsymmetrical 3,4-disubstituted maleimides based on transition-metal catalysts
F. Bandehali-Naeini, Z. Tanbakouchian, N. Farajinia-Lehi, N. Mayer, M. Shiri,* M. Breugst,* Org. Biomol. Chem., 2024, 22, 380–387.
DOI: 10.1039/D3OB01620J
F. Bandehali-Naeini, Z. Tanbakouchian, N. Farajinia-Lehi, N. Mayer, M. Shiri,* M. Breugst,* Org. Biomol. Chem., 2024, 22, 380–387.
DOI: 10.1039/D3OB01620J

71. Trendberichte Organische Chemie 2024
M. Breugst,* J. Andexer, L. Barra, S. B. Beil, R. Breinbauer, I. Burkhardt, O. Dumele, M. Ernst, U. Gellrich, P. Germer, M. Giese, P. Huy, S. Kath-Schorr, J. Klepp, K. Körber, M. Kordes, C. A. Kuttruff, T. Lindel, S. Myllek, F. Pfrengle, J. Pietruszka, N. Schaschke, M. O. Senge, G. Storch, J. F. Teichert, J. Tönjes, S. R. Waldvogel, T. Werner, C. Winter Nachr. Chem., 2024, 72, 44–67.
DOI: 10.1002/nadc.20244139258
M. Breugst,* J. Andexer, L. Barra, S. B. Beil, R. Breinbauer, I. Burkhardt, O. Dumele, M. Ernst, U. Gellrich, P. Germer, M. Giese, P. Huy, S. Kath-Schorr, J. Klepp, K. Körber, M. Kordes, C. A. Kuttruff, T. Lindel, S. Myllek, F. Pfrengle, J. Pietruszka, N. Schaschke, M. O. Senge, G. Storch, J. F. Teichert, J. Tönjes, S. R. Waldvogel, T. Werner, C. Winter Nachr. Chem., 2024, 72, 44–67.
DOI: 10.1002/nadc.20244139258
2023
70. Concepts and Practical Aspects of Computational Chemistry (Book Chapter)
M. Breugst, in Enabling Tools and Techniques for Organic Synthesis (Ed. S. G. Newman), 2023, 259–311.
DOI: 10.1002/9781119855668.ch7
ISBN (print): 978-1-119-85563-7
M. Breugst, in Enabling Tools and Techniques for Organic Synthesis (Ed. S. G. Newman), 2023, 259–311.
DOI: 10.1002/9781119855668.ch7
ISBN (print): 978-1-119-85563-7

69. Divergent synthesis of pyrrole carboxamides from pyrrole carboxaldehyde and formamides /amines via oxidative amidation involving pyrrole acyl radicals
J. K. Laha,* S. Panday, J. P. Weber, M. Breugst,* Chem. Commun., 2023, 60, 10259–10262.
DOI: 10.1039/D3CC02766J
J. K. Laha,* S. Panday, J. P. Weber, M. Breugst,* Chem. Commun., 2023, 60, 10259–10262.
DOI: 10.1039/D3CC02766J

68. Transition Metal-Free Regioselective Phosphonation of Pyridines: Scope and Mechanism
V. Quint, T. H. V. Nguyen, G. Mathieu, S. Chelli, M. Breugst,* J.-F. Lohier, A.-C. Gaumont,S. Lakhdar,* ACS Org. Inorg. Au, 2023, 3, 151–157.
DOI: 10.1021/acsorginorgau.2c00055
V. Quint, T. H. V. Nguyen, G. Mathieu, S. Chelli, M. Breugst,* J.-F. Lohier, A.-C. Gaumont,S. Lakhdar,* ACS Org. Inorg. Au, 2023, 3, 151–157.
DOI: 10.1021/acsorginorgau.2c00055

67. Iodine-Catalyzed Claisen-Rearrangements of Allyl Aryl Ethers and Subsequent Iodocyclizations
T. Arndt, A. Raina, M. Breugst,* Chem. Asian J., 2023, 18, e202201279.
DOI: 10.1002/asia.202201279
Invited contribution to the special collection Halogen Bonding.
T. Arndt, A. Raina, M. Breugst,* Chem. Asian J., 2023, 18, e202201279.
DOI: 10.1002/asia.202201279
Invited contribution to the special collection Halogen Bonding.

66. Trendberichte Organische Chemie 2023
M. Breugst,* J. N. Andexer, S. B. Beil, R. Breinbauer, O. Dumele, M. Ernst, U. Gellrich, P. Germer, M. Giese, T. A. M. Gulder, P. Huy, W. Hüttel, S. Kath-Schorr, K. Körber, M. Kordes, C. Kuttruff, T. Lindel, R. Meier, S. Myllek, N. Schaschke, F. Pfrengle, J. Pietruszka, H. Sebode, M. O. Senge, G. Storch, B. F. Straub, J. Teichert, S. R. Waldvogel, T. Werner, C. Winter Nachr. Chem., 2023, 71, 40–66.
DOI: 10.1002/nadc.20234135542
M. Breugst,* J. N. Andexer, S. B. Beil, R. Breinbauer, O. Dumele, M. Ernst, U. Gellrich, P. Germer, M. Giese, T. A. M. Gulder, P. Huy, W. Hüttel, S. Kath-Schorr, K. Körber, M. Kordes, C. Kuttruff, T. Lindel, R. Meier, S. Myllek, N. Schaschke, F. Pfrengle, J. Pietruszka, H. Sebode, M. O. Senge, G. Storch, B. F. Straub, J. Teichert, S. R. Waldvogel, T. Werner, C. Winter Nachr. Chem., 2023, 71, 40–66.
DOI: 10.1002/nadc.20234135542
2022
65. TBHP-Mediated Denitrogenative Synthesis of Pyridine Carboxamides from Pyridine Carbohydrazides and Amines in Water
J. K. Laha,* A. Gupta, U. Gulati, M. K. Hunjan, J. P. Weber, M. Breugst,* Org. Chem. Front., 2022, 9, 6902–5908.
DOI: 10.1039/D2QO01465C
J. K. Laha,* A. Gupta, U. Gulati, M. K. Hunjan, J. P. Weber, M. Breugst,* Org. Chem. Front., 2022, 9, 6902–5908.
DOI: 10.1039/D2QO01465C

64. Stereoselective Synthesis of 2-Oxyenamides
S.-C. Krieg, J. Grimmer, A. M. Pick, H. Kelm, M. Breugst, G. Manolikakes, Eur. J. Org. Chem., 2022, e202200772.
DOI: 10.1002/ejoc.202200772
S.-C. Krieg, J. Grimmer, A. M. Pick, H. Kelm, M. Breugst, G. Manolikakes, Eur. J. Org. Chem., 2022, e202200772.
DOI: 10.1002/ejoc.202200772

63. pH-Controlled Intramolecular Decarboxylative Cyclization of Biarylacetic Acids: Implication on Umpolung Reactivity of Aroyl Radicals
J. K. Laha,* U. Gulati, Saima, T. Schulte, M. Breugst,* J. Org. Chem., 2022, 87, 6638–6656.
DOI: 10.1021/acs.joc.2c00295
J. K. Laha,* U. Gulati, Saima, T. Schulte, M. Breugst,* J. Org. Chem., 2022, 87, 6638–6656.
DOI: 10.1021/acs.joc.2c00295

62. Formation of Breslow Intermediates from N-Heterocyclic Carbenes and Aldehydes Involves Autocatalysis by the Breslow Intermediate, and a Hemiacetal
A. Wessels, M. Klußmann, M. Breugst, N. E. Schlörer, A. Berkessel, Angew. Chem. Int. Ed., 2022, 61, e202117682; Angew. Chem., 2022, 132, e202117682.
DOI: 10.1002/anie.202117682
Selected as a Hot Paper.
A. Wessels, M. Klußmann, M. Breugst, N. E. Schlörer, A. Berkessel, Angew. Chem. Int. Ed., 2022, 61, e202117682; Angew. Chem., 2022, 132, e202117682.
DOI: 10.1002/anie.202117682
Selected as a Hot Paper.

61. Ammonium Pertechnetate in Mixtures of Trifluoromethanesulfonic Acid and Trifluoromethanesulfonic Anhydride
M. Zegke,* D. Grödler, M. Roca Jungfer, A. Haseloer, M. Kreuter, J. M. Neudörfl, T. Sittel, C. M. James, J. Rothe, M. Altmaier, A. Klein, M. Breugst,* U. Abram, E. Strub,* M. S. Wickleder,* Angew. Chem. Int. Ed., 2022, 61, e202113777; Angew. Chem., 2022, 134, e202113777.
DOI: 10.1002/anie.202113777
Selected as a Hot Paper.
M. Zegke,* D. Grödler, M. Roca Jungfer, A. Haseloer, M. Kreuter, J. M. Neudörfl, T. Sittel, C. M. James, J. Rothe, M. Altmaier, A. Klein, M. Breugst,* U. Abram, E. Strub,* M. S. Wickleder,* Angew. Chem. Int. Ed., 2022, 61, e202113777; Angew. Chem., 2022, 134, e202113777.
DOI: 10.1002/anie.202113777
Selected as a Hot Paper.

60. Trendberichte Organische Chemie 2022
J. N. Andexer, U. Beifuss, M. Brasholz, R. Breinbauer, M. Breugst,* O. Dumele, M. Ernst, R. Ganardi, M. Giese, T. A. M. Gulder, W. Hüttel, S. Kath-Schorr, K. Körber, M. Kordes, T. Lindel, C. Mück-Lichtenfeld, J. Niemeyer, R. Pfau, F. Pfrengle, J. Pietruszka, J. L. Röckl, N. Schaschke, H. Sebode, M. O. Senge, B. F. Straub, J. Teichert, S. R. Waldvogel, T. Werner, C. Winter Nachr. Chem., 2022, 70, 42–697.
DOI: 10.1002/nadc.20224122453
J. N. Andexer, U. Beifuss, M. Brasholz, R. Breinbauer, M. Breugst,* O. Dumele, M. Ernst, R. Ganardi, M. Giese, T. A. M. Gulder, W. Hüttel, S. Kath-Schorr, K. Körber, M. Kordes, T. Lindel, C. Mück-Lichtenfeld, J. Niemeyer, R. Pfau, F. Pfrengle, J. Pietruszka, J. L. Röckl, N. Schaschke, H. Sebode, M. O. Senge, B. F. Straub, J. Teichert, S. R. Waldvogel, T. Werner, C. Winter Nachr. Chem., 2022, 70, 42–697.
DOI: 10.1002/nadc.20224122453
2021
59. Enantioselective Rauhut–Currier Reaction with β-Substituted Acrylamides Catalyzed by N-Heterocyclic Carbenes
V. Pitchumani, M. Breugst, D. W. Lupton, Org. Lett., 2021, 23, 9413–9418.
DOI: 10.1021/acs.orglett.1c03554
V. Pitchumani, M. Breugst, D. W. Lupton, Org. Lett., 2021, 23, 9413–9418.
DOI: 10.1021/acs.orglett.1c03554

58. Some Surprising Transformations of Colchicone and other Colchicine-derived Tropolones
A., P. Hilken nee Thomopoulou, T. Schulte, J.-M. Neudörfl, M. Breugst,* H.-G. Schmalz,* Eur. J. Org. Chem., 2021, 6375–6382.
DOI: 10.1002/ejoc.202100999
Selected as a VIP Paper.
A., P. Hilken nee Thomopoulou, T. Schulte, J.-M. Neudörfl, M. Breugst,* H.-G. Schmalz,* Eur. J. Org. Chem., 2021, 6375–6382.
DOI: 10.1002/ejoc.202100999
Selected as a VIP Paper.

57. Iodine‐Catalyzed Diels‐Alder Reactions
T. Arndt, P. K. Wagner, J. J. Koenig, M. Breugst,* ChemCatChem, 2021, 13, 2922–2930.
DOI: 10.1002/cctc.202100342
Invited contribution to the special collection Young Researchers Series
T. Arndt, P. K. Wagner, J. J. Koenig, M. Breugst,* ChemCatChem, 2021, 13, 2922–2930.
DOI: 10.1002/cctc.202100342
Invited contribution to the special collection Young Researchers Series

56. Catalysis by Molecular Iodine (Book Chapter)
J. J. König, M. Breugst,* in Halogen Bonding in Solution (Ed. S. M. Huber), 2021, 233–268.
DOI: 10.1002/9783527825738.ch7
ISBN (print): 978-3-527-34731-5
J. J. König, M. Breugst,* in Halogen Bonding in Solution (Ed. S. M. Huber), 2021, 233–268.
DOI: 10.1002/9783527825738.ch7
ISBN (print): 978-3-527-34731-5

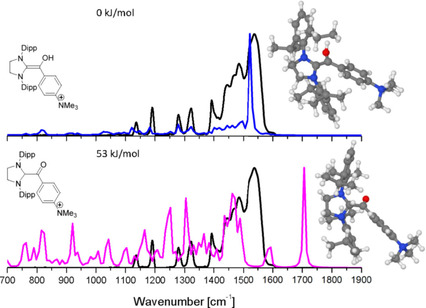
55. Breslow Intermediates (Aminoenols) and their Keto Tautomers: First Gas‐Phase Characterization by IR Ion Spectroscopy
M. Paul, K. Peckelsen, T. Thomulka, J. Martens, G. Berden, J. Oomens, J. M. Neudörfl, M. Breugst,* A. J. H. M. Meijer,* M. Schäfer,* A. Berkessel,* Chem. Eur. J., 2021, 27, 2662–2669.
DOI: 10.1002/chem.202003454
Selected as a VIP Paper
M. Paul, K. Peckelsen, T. Thomulka, J. Martens, G. Berden, J. Oomens, J. M. Neudörfl, M. Breugst,* A. J. H. M. Meijer,* M. Schäfer,* A. Berkessel,* Chem. Eur. J., 2021, 27, 2662–2669.
DOI: 10.1002/chem.202003454
Selected as a VIP Paper
2020
54. Enantioselective N-Heterocyclic Carbene Catalysis via the Acyl Azolium without Exogenous Oxidants
J. Cao, R. Gillard, A. Jahanbakhsh, M. Breugst, D. W. Lupton, ACS Catal., 2020, 10, 11791–11796.
DOI: 10.1021/acscatal.0c02705
J. Cao, R. Gillard, A. Jahanbakhsh, M. Breugst, D. W. Lupton, ACS Catal., 2020, 10, 11791–11796.
DOI: 10.1021/acscatal.0c02705

53. Competition Between N and O: Use of Diazine N-Oxides as a Test Case for the Marcus Theory Rationale for Ambident Reactivity
K. Sheehy, L. M. Bateman, N. T. Flosbach, M. Breugst,* P. A. Byrne,* Chem. Sci., 2020, 11, 9630–9647.
DOI: 10.1039/D0SC02834G
K. Sheehy, L. M. Bateman, N. T. Flosbach, M. Breugst,* P. A. Byrne,* Chem. Sci., 2020, 11, 9630–9647.
DOI: 10.1039/D0SC02834G

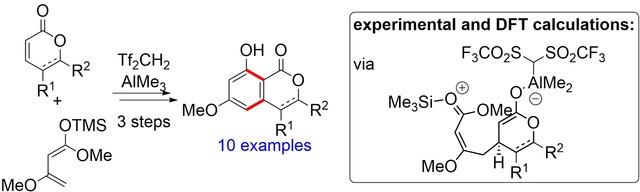
52. Experimental and Computational Investigations of the Reaction between α,β‐unsaturated Lactones and 1,3‐Dienes by Cooperative Lewis‐Acid/Brønsted‐Acid Catalysis
A. Weber, M. Breugst,* J. Pietruszka,* Angew. Chem. Int. Ed., 2020, 59, 18709–18716; Angew. Chem., 2020, 132, 18868–18875.
DOI: 10.1002/anie.202008365
A. Weber, M. Breugst,* J. Pietruszka,* Angew. Chem. Int. Ed., 2020, 59, 18709–18716; Angew. Chem., 2020, 132, 18868–18875.
DOI: 10.1002/anie.202008365
51. σ‐Hole Interactions in Catalysis
M. Breugst,* J. J. König, Eur. J. Org. Chem., 2020, 5473–5487.
DOI: 10.1002/ejoc.202000660
Selected as a VIP Paper. Also included in the virtual issue: Hot Topic: Organocatalysis
M. Breugst,* J. J. König, Eur. J. Org. Chem., 2020, 5473–5487.
DOI: 10.1002/ejoc.202000660
Selected as a VIP Paper. Also included in the virtual issue: Hot Topic: Organocatalysis

50. The Huisgen Reaction – Milestones of the 1,3-Dipolar Cycloaddition
M. Breugst,* H. Reißig,* Angew. Chem. Int. Ed., 2020, 59, 12293–12307; Angew. Chem., 2020, 132, 12389–12404.
DOI: 10.1002/anie.202003115
Also included in the virtual issues: Hot Topic: Click Chemistry and In memory of Rolf Huisgen
M. Breugst,* H. Reißig,* Angew. Chem. Int. Ed., 2020, 59, 12293–12307; Angew. Chem., 2020, 132, 12389–12404.
DOI: 10.1002/anie.202003115
Also included in the virtual issues: Hot Topic: Click Chemistry and In memory of Rolf Huisgen

49. Unambiguous Identification of N- or O-Alkylation of Aromatic Nitrogen Heterocycles and N-Oxides Using 1H-15N HMBC NMR Spectroscopy
K. J. Sheehy, L. M. Bateman, N. T. Flosbach, M. Breugst,* P. A. Byrne,* Eur. J. Org. Chem., 2020, 3270–3281.
DOI: 10.1002/ejoc.202000329
Also included in the virtual issue: In memory of Rolf Huisgen
K. J. Sheehy, L. M. Bateman, N. T. Flosbach, M. Breugst,* P. A. Byrne,* Eur. J. Org. Chem., 2020, 3270–3281.
DOI: 10.1002/ejoc.202000329
Also included in the virtual issue: In memory of Rolf Huisgen

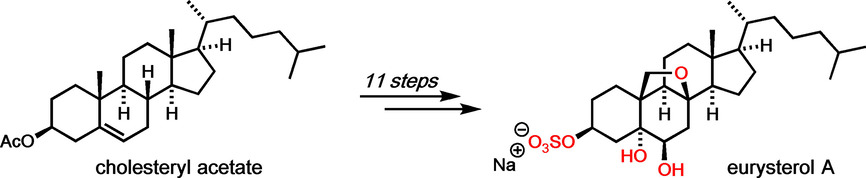
48. Synthesis of the 8,19‐Epoxysteroid Eurysterol A
Ö. Taspinar, T. Wilczek, J. Erver, M. Breugst, J.-M. Neudörfl, H.-G. Schmalz, Chem. Eur. J., 2020, 26, 4256–4260.
DOI: 10.1002/chem.202000585
Ö. Taspinar, T. Wilczek, J. Erver, M. Breugst, J.-M. Neudörfl, H.-G. Schmalz, Chem. Eur. J., 2020, 26, 4256–4260.
DOI: 10.1002/chem.202000585
47. Enantioselective Allylation of Indoles: A Surprising Diastereoselectivity
P. Ullrich, J. Schmauck, M. Brauns, M. Mantel, M. Breugst,* J. Pietruszka,* J. Org. Chem., 2020, 85, 1894–1905.
DOI: 10.1021/acs.joc.9b02573
P. Ullrich, J. Schmauck, M. Brauns, M. Mantel, M. Breugst,* J. Pietruszka,* J. Org. Chem., 2020, 85, 1894–1905.
DOI: 10.1021/acs.joc.9b02573

2019
46. The Technical Synthesis of 1,5,9-Cyclododecatriene Revisited: Surprising By-products from a Venerable Industrial Process
F. Thrun, V. Hickmann, C. Stock, A. Schaefer, W. Maier, M. Breugst, N. E. Schlörer, A. Berkessel, H. Teles, J. Org. Chem., 2019, 84, 13211–13220.
DOI: 10.1021/acs.joc.9b01633
Selected as a Featured Article.
F. Thrun, V. Hickmann, C. Stock, A. Schaefer, W. Maier, M. Breugst, N. E. Schlörer, A. Berkessel, H. Teles, J. Org. Chem., 2019, 84, 13211–13220.
DOI: 10.1021/acs.joc.9b01633
Selected as a Featured Article.

45. N-Heterocyclic Carbene Catalyzed (5 + 1) Annulations Exploiting a Vinyl Dianion Synthon Strategy
X. B. Nguyen, Y. Nakano, N. M. Duggan, L. Scott, M. Breugst, D. W. Lupton, Angew. Chem. Int. Ed., 2019, 58, 11483–11490; Angew. Chem., 2019, 131, 11607–11614.
DOI: 10.1002/anie.201905475
X. B. Nguyen, Y. Nakano, N. M. Duggan, L. Scott, M. Breugst, D. W. Lupton, Angew. Chem. Int. Ed., 2019, 58, 11483–11490; Angew. Chem., 2019, 131, 11607–11614.
DOI: 10.1002/anie.201905475

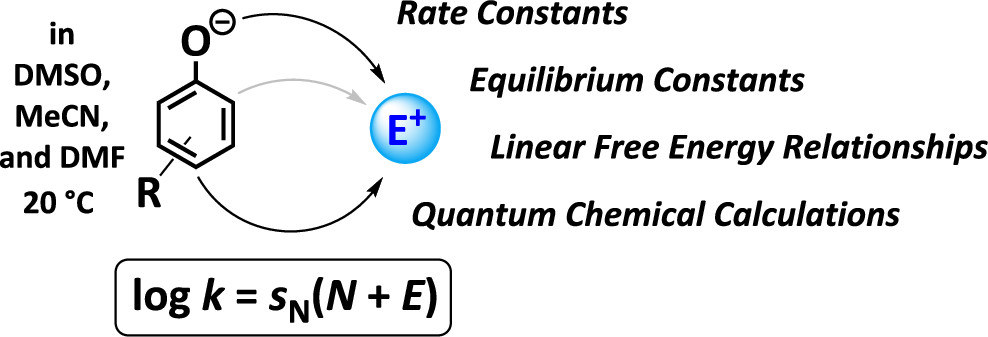
44. Ambident Reactivity of Phenolate Anions Revisited: A Quantitative Approach to Phenolate Reactivities
R. J. Mayer, M. Breugst, N. Hampel, A. R. Ofial, H. Mayr, J. Org. Chem., 2019, 84, 8837–8858.
DOI: 10.1021/acs.joc.9b01485
Selected as a Featured Article.
R. J. Mayer, M. Breugst, N. Hampel, A. R. Ofial, H. Mayr, J. Org. Chem., 2019, 84, 8837–8858.
DOI: 10.1021/acs.joc.9b01485
Selected as a Featured Article.
43. Iodine-Catalyzed Nazarov Cyclizations
J. J. König, T. Arndt, N. Gildemeister, J.-M. Neudörfl, M. Breugst*, J. Org. Chem., 2019, 84, 7587–7605.
DOI: 10.1021/acs.joc.9b01083
Selected as a Featured Article.
J. J. König, T. Arndt, N. Gildemeister, J.-M. Neudörfl, M. Breugst*, J. Org. Chem., 2019, 84, 7587–7605.
DOI: 10.1021/acs.joc.9b01083
Selected as a Featured Article.

42. Philicity of Acetonyl and Benzoyl Radicals: a Comparative Experimental and Computational Study
R. H. Verschueren, J. Schmauck, M. S. Perryman, H.-L. Yue, J. Riegger, B. Schweitzer-Chaput, M. Breugst,* M. Klussmann,* Chem. Eur. J., 2019, 25, 9088–9097.
DOI: 10.1002/chem.201901439
Selected as a Hot Paper.
R. H. Verschueren, J. Schmauck, M. S. Perryman, H.-L. Yue, J. Riegger, B. Schweitzer-Chaput, M. Breugst,* M. Klussmann,* Chem. Eur. J., 2019, 25, 9088–9097.
DOI: 10.1002/chem.201901439
Selected as a Hot Paper.

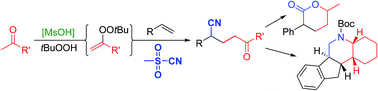
41. Radical Addition of Ketones and Cyanide to Olefins via Acid Catalyzed Formation of Intermediate Alkenyl Peroxides
W. Shao, M. Lux, M. Breugst, M. Klussmann, Org. Chem. Front., 2019, 6, 1796–1800.
DOI: 10.1039/C9QO00447E
W. Shao, M. Lux, M. Breugst, M. Klussmann, Org. Chem. Front., 2019, 6, 1796–1800.
DOI: 10.1039/C9QO00447E
40. Intermediates of N‐Heterocyclic Carbene (NHC) Dimerization Probed in the Gas Phase by Ion Mobility Mass Spectrometry: C‐H⋯:C Hydrogen Bonding vs. Covalent Dimer Formation
M. Paul,* E. Detmar, M. Schlangen, M. Breugst,* J.-M. Neudörfl, H. Schwarz, A. Berkessel,* M. Schäfer,* Chem. Eur. J., 2019, 67, 2511–2518.
DOI: 10.1002/chem.201803641
M. Paul,* E. Detmar, M. Schlangen, M. Breugst,* J.-M. Neudörfl, H. Schwarz, A. Berkessel,* M. Schäfer,* Chem. Eur. J., 2019, 67, 2511–2518.
DOI: 10.1002/chem.201803641

39. Carbonyl-Olefin Metathesis Catalyzed by Molecular Iodine
U. P. N. Tran, G. Oss, M. Breugst, E. Detmar, D. P. Pace, K. Liyanto, T. V. Nguyen, ACS Catal., 2019, 9, 912–919.
DOI: 10.1021/acscatal.8b03769
U. P. N. Tran, G. Oss, M. Breugst, E. Detmar, D. P. Pace, K. Liyanto, T. V. Nguyen, ACS Catal., 2019, 9, 912–919.
DOI: 10.1021/acscatal.8b03769

38. Reaktionen im kontinuierlichen Strom
J. König, M. Breugst,* Nachr. Chem., 2019, 67, 81–85.
DOI: 10.1002/nadc.20194084228
J. König, M. Breugst,* Nachr. Chem., 2019, 67, 81–85.
DOI: 10.1002/nadc.20194084228

2018
37. Katalyse mit elektrophilen Phosphoniumionen
J. Schmauck, M. Breugst,* Nachr. Chem., 2018, 66, 862–865.
DOI: 10.1002/nadc.20184077309
J. Schmauck, M. Breugst,* Nachr. Chem., 2018, 66, 862–865.
DOI: 10.1002/nadc.20184077309

36. Redetermination of the Solvent-Free Crystal Structure of L-Proline
J. J. König, J.-M. Neudörfl, A. Hansen, M. Breugst,* Acta Crystallogr., Sect. E: Crystallogr. Commun., 2018, E74, 1067–1070.
DOI: 10.1107/S2056989018009490
J. J. König, J.-M. Neudörfl, A. Hansen, M. Breugst,* Acta Crystallogr., Sect. E: Crystallogr. Commun., 2018, E74, 1067–1070.
DOI: 10.1107/S2056989018009490

35. Visible Light–Mediated Metal–Free Synthesis of Aryl Phosphonates: Synthetic and Mechanistic Investigations
W. Lecroq, P. Bazille, F. Morlet-Savary, J. Lalevée, M. Breugst,* A.-C. Gaumont, S. Lakhdar,* Org. Lett., 2018, 20, 4164–4167.
DOI: 10.1021/acs.orglett.8b01379
selected for the Organic Letters Global Enterprise Virtual Issue
highlighted on www.organic-chemistry.org: link
W. Lecroq, P. Bazille, F. Morlet-Savary, J. Lalevée, M. Breugst,* A.-C. Gaumont, S. Lakhdar,* Org. Lett., 2018, 20, 4164–4167.
DOI: 10.1021/acs.orglett.8b01379
selected for the Organic Letters Global Enterprise Virtual Issue
highlighted on www.organic-chemistry.org: link

34. Cobalt-Catalyzed C–H Cyanations: Insights into the Reaction Mechanism and the Role of London Dispersion
E. Detmar, V. Müller, D. Zell, L. Ackermann,* M. Breugst,* Beilstein J. Org. Chem., 2018, 14, 1537–1545.
DOI: 10.3762/bjoc.14.130
Invited contribution to the Thematic Series Dispersion interactions.
E. Detmar, V. Müller, D. Zell, L. Ackermann,* M. Breugst,* Beilstein J. Org. Chem., 2018, 14, 1537–1545.
DOI: 10.3762/bjoc.14.130
Invited contribution to the Thematic Series Dispersion interactions.
33. Sperrige Substituenten – Zwischen Attraktion und Repulsion
J. J. König, M. Breugst,* Nachr. Chem., 2018, 66, 505–509.
DOI: 10.1002/nadc.20184073225
J. J. König, M. Breugst,* Nachr. Chem., 2018, 66, 505–509.
DOI: 10.1002/nadc.20184073225

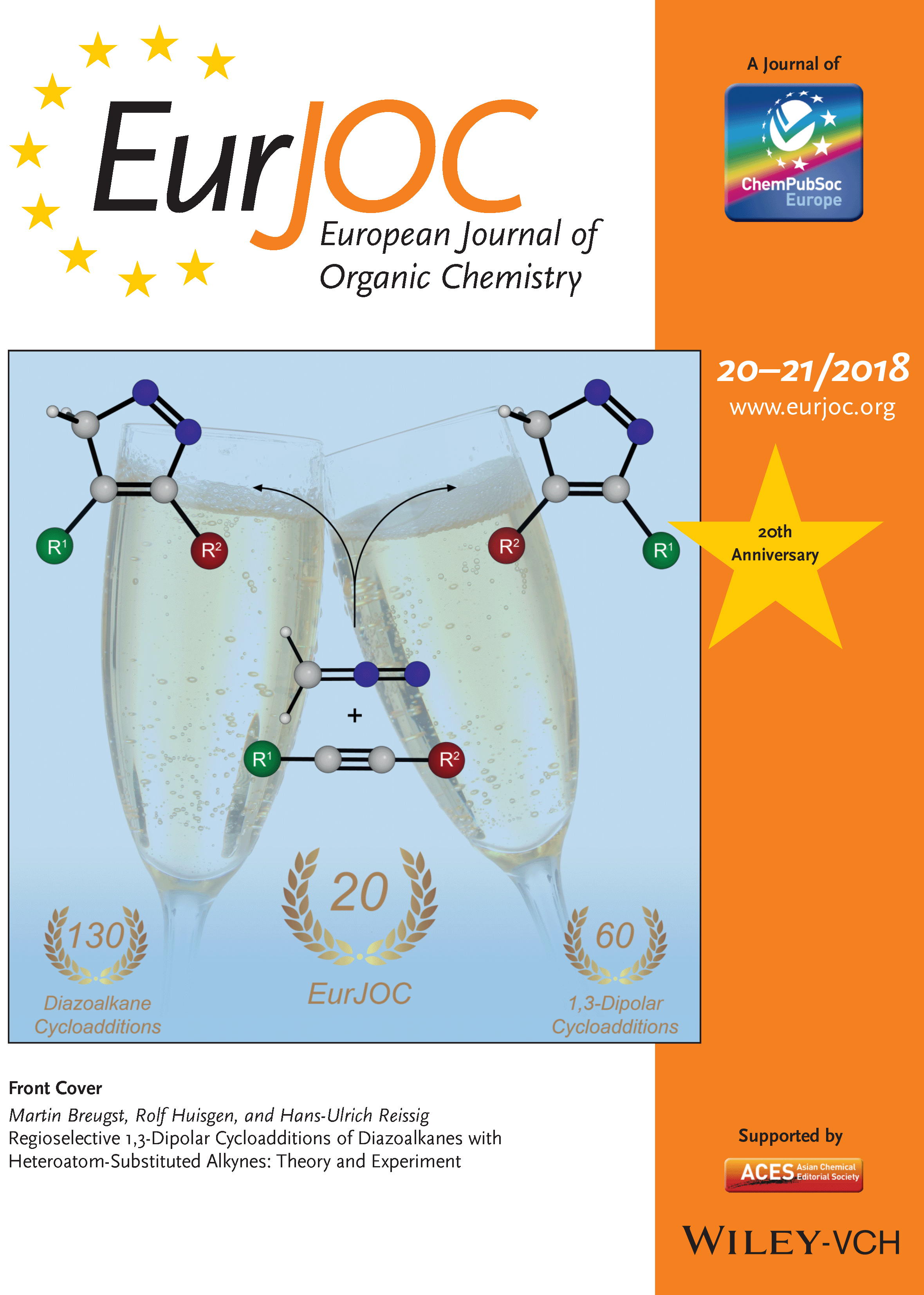
32. Regioselective 1,3-Dipolar Cycloadditions of Diazoalkanes with Heteroatom-Substituted Alkynes: Theory and Experiment
M. Breugst,* R. Huisgen, H.-U. Reissig,* Eur. J. Org. Chem., 2018, 2477–2485.
DOI: 10.1002/ejoc.201800100
invited contribution for the 20th Anniversary (Celebrating the Past, Present and Future)
highlighted in a ChemViews Magazine interview with the authors
M. Breugst,* R. Huisgen, H.-U. Reissig,* Eur. J. Org. Chem., 2018, 2477–2485.
DOI: 10.1002/ejoc.201800100
invited contribution for the 20th Anniversary (Celebrating the Past, Present and Future)
highlighted in a ChemViews Magazine interview with the authors

31. Mechanisms in Iodine Catalysis
M. Breugst,* D. von der Heiden, Chem. Eur. J., 2018, 24, 9187–9199.
DOI: 10.1002/chem.201706136
Selected by the Editorial Office for the Showcase of outstanding Review-type articles.
M. Breugst,* D. von der Heiden, Chem. Eur. J., 2018, 24, 9187–9199.
DOI: 10.1002/chem.201706136
Selected by the Editorial Office for the Showcase of outstanding Review-type articles.

30. Activation of Michael Acceptors by Halogen-Bond Donors
D. von der Heiden, E. Detmar, R. Kuchta, M. Breugst,* Synlett, 2018, 1307–1313.
DOI: 10.1055/s-0036-1591841
invited contribution for the Special Section 9th EuCheMS Organic Division Young Investigator Workshop
D. von der Heiden, E. Detmar, R. Kuchta, M. Breugst,* Synlett, 2018, 1307–1313.
DOI: 10.1055/s-0036-1591841
invited contribution for the Special Section 9th EuCheMS Organic Division Young Investigator Workshop

2017
29. The potential of pnicogen bonding for catalysis – A computational study
J. Schmauck, M. Breugst,* Org. Biomol. Chem., 2017, 15, 8037–8045.
DOI: 10.1039/C7OB01599B
J. Schmauck, M. Breugst,* Org. Biomol. Chem., 2017, 15, 8037–8045.
DOI: 10.1039/C7OB01599B

28. Kinetics of Electrophilic Alkylations of Barbiturate and Thiobarbiturate Anions
A. Schade, I. Tchernook, M. Bauer, A. Oehlke, M. Breugst, J. Friedrich, S. Spange, J. Org. Chem., 2017, 82, 8476–8488.
DOI: 10.1021/acs.joc.7b01223
A. Schade, I. Tchernook, M. Bauer, A. Oehlke, M. Breugst, J. Friedrich, S. Spange, J. Org. Chem., 2017, 82, 8476–8488.
DOI: 10.1021/acs.joc.7b01223
27. Novel Noncovalent Interactions in Catalysis: A Focus on Halogen, Chalcogen, and Anion-π Bonding
M. Breugst,* D. von der Heiden, J. Schmauck, Synthesis, 2017, 49, 3224–3236.
DOI: 10.1055/s-0036-1588838
M. Breugst,* D. von der Heiden, J. Schmauck, Synthesis, 2017, 49, 3224–3236.
DOI: 10.1055/s-0036-1588838

26. Highly Enantioselective Allylation of Ketones - An Efficient Approach to all Stereoisomers of Tertiary Homoallylic Alcohols
M. Brauns, M. Mantel, J. Schmauck, M. Guder, M. Breugst,* J. Pietruszka,* Chem. Eur. J., 2017, 23, 12136–12140.
DOI: 10.1002/chem.201701740
M. Brauns, M. Mantel, J. Schmauck, M. Guder, M. Breugst,* J. Pietruszka,* Chem. Eur. J., 2017, 23, 12136–12140.
DOI: 10.1002/chem.201701740

25. Reaction Mechanism of Iodine-Catalyzed Michael Additions
D. von der Heiden, S. Bozkus, M. Klussmann, M. Breugst,* J. Org. Chem. 2017, 82, 4037–4043.
DOI: 10.1021/acs.joc.7b00445
Selected as a Featured Article.
D. von der Heiden, S. Bozkus, M. Klussmann, M. Breugst,* J. Org. Chem. 2017, 82, 4037–4043.
DOI: 10.1021/acs.joc.7b00445
Selected as a Featured Article.

2016
24. A Metal–Free Synthesis of 6–Phosphorylated Phenanthridines: Synthetic and Mechanistic Insights
L. Noël-Duchesneau, E. Lagadic, F. Morlet-Savary, J.-F. Lohier, I. Chataigner, M. Breugst,* J. Lalevée, A.-C. Gaumont, S. Lakhdar,* Org. Lett. 2016, 18, 5900–5903.
DOI: 10.1021/acs.orglett.6b02983
Highlighted in Synfacts 2017, 13, 140.
L. Noël-Duchesneau, E. Lagadic, F. Morlet-Savary, J.-F. Lohier, I. Chataigner, M. Breugst,* J. Lalevée, A.-C. Gaumont, S. Lakhdar,* Org. Lett. 2016, 18, 5900–5903.
DOI: 10.1021/acs.orglett.6b02983
Highlighted in Synfacts 2017, 13, 140.

23. Redox-Neutral Aromatization of Cyclic Amines: Mechanistic Insights and Harnessing of Reactive Intermediates for Amine α- and β-C-H Functionalization
L. Ma, A. Paul, M. Breugst,* D. Seidel,* Chem. Eur. J. 2016, 22, 18179–18189.
DOI: 10.1002/chem.201603839
L. Ma, A. Paul, M. Breugst,* D. Seidel,* Chem. Eur. J. 2016, 22, 18179–18189.
DOI: 10.1002/chem.201603839

22. Influence of the N-Substituents on the Nucleophilicity and Lewis Basicity of N-Heterocyclic Carbenes
A. Levens, F. An, M. Breugst, H. Mayr, D. W. Lupton, Org. Lett. 2016, 18, 3566–3569.
DOI: 10.1021/acs.orglett.6b01525
A. Levens, F. An, M. Breugst, H. Mayr, D. W. Lupton, Org. Lett. 2016, 18, 3566–3569.
DOI: 10.1021/acs.orglett.6b01525

21. Origin of the Catalytic Effects of Molecular Iodine – A Computational Analysis
M. Breugst,* E. Detmar, D. von der Heiden, ACS Catal. 2016, 6, 3203–3212.
DOI: 10.1021/acscatal.6b00447
M. Breugst,* E. Detmar, D. von der Heiden, ACS Catal. 2016, 6, 3203–3212.
DOI: 10.1021/acscatal.6b00447
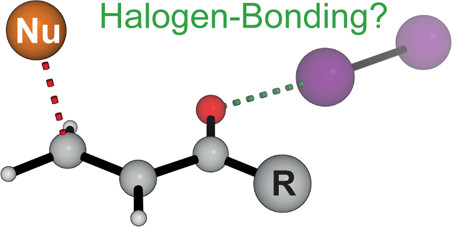
20. Quantification of the Nucleophilic Reactivity of Nicotine
P. A. Byrne, S. Kobayashi, M. Breugst, H. Laub, H. Mayr, J. Phys. Org. Chem. 2016, 29, 759–767.
DOI: 10.1002/poc.3580
P. A. Byrne, S. Kobayashi, M. Breugst, H. Laub, H. Mayr, J. Phys. Org. Chem. 2016, 29, 759–767.
DOI: 10.1002/poc.3580

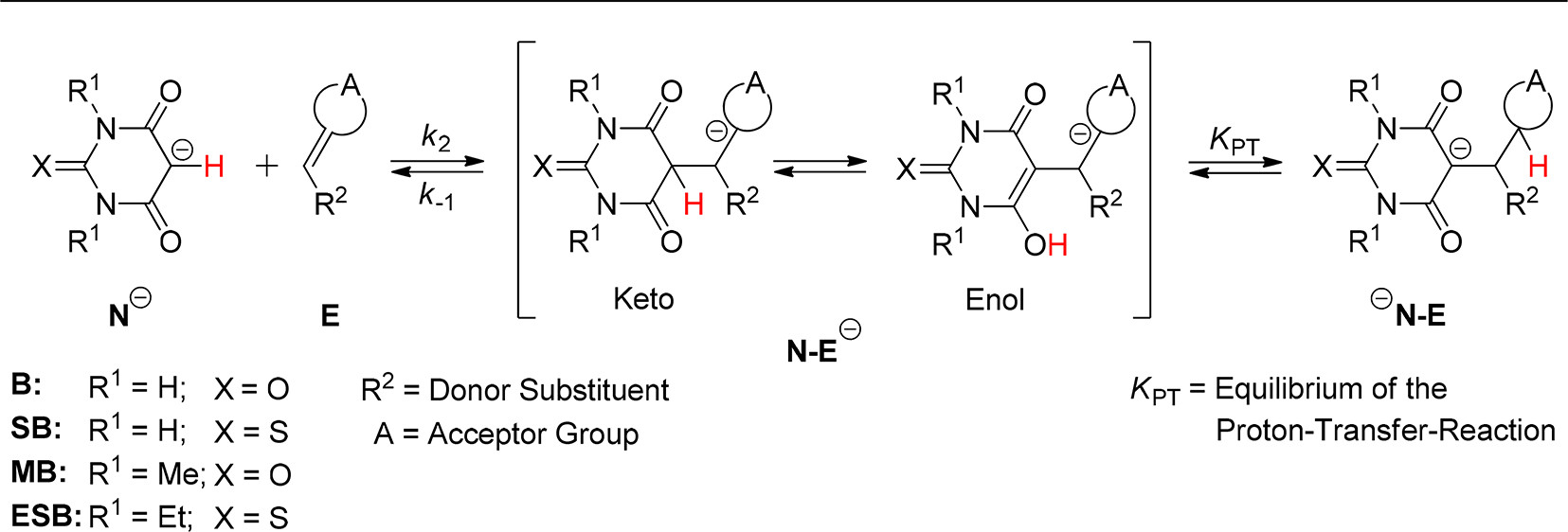
19. Keto-Enol Thermodynamics of Breslow Intermediates
M. Paul, M. Breugst, J.-M. Neudörfl, R. B. Sunoj, A. Berkessel, J. Am. Chem. Soc. 2016, 138, 5044–5051.
DOI: 10.1021/jacs.5b13236
M. Paul, M. Breugst, J.-M. Neudörfl, R. B. Sunoj, A. Berkessel, J. Am. Chem. Soc. 2016, 138, 5044–5051.
DOI: 10.1021/jacs.5b13236

18. Enantioselective Catalysts for the Synthesis of α-Substituted Allyboronates - An Accelerated Approach towards Isomerically Pure Homoallylic Alcohols
M. Brauns, F. Muller, D. Gülden, D. Böse, W. Frey, M. Breugst,* J. Pietruszka,* Angew. Chem. Int. Ed. 2016, 55, 1548–1552; Angew. Chem. 2016, 128, 1574–1578.
DOI: 10.1002/anie.201509198
M. Brauns, F. Muller, D. Gülden, D. Böse, W. Frey, M. Breugst,* J. Pietruszka,* Angew. Chem. Int. Ed. 2016, 55, 1548–1552; Angew. Chem. 2016, 128, 1574–1578.
DOI: 10.1002/anie.201509198

2015
17. Mit molekularem Iod katalysieren
M. Breugst, Nachr. Chem. 2015, 63, 1180–1183.
DOI: 10.1002/nadc.201590404
M. Breugst, Nachr. Chem. 2015, 63, 1180–1183.
DOI: 10.1002/nadc.201590404

16. Asymmetric Redox-Annulation of Cyclic Amines
Y. Kang, W. Chen, M. Breugst,* D. Seidel,* J. Org. Chem. 2015, 80, 9628–9640.
DOI: 10.1021/acs.joc.5b01384
Y. Kang, W. Chen, M. Breugst,* D. Seidel,* J. Org. Chem. 2015, 80, 9628–9640.
DOI: 10.1021/acs.joc.5b01384

15. Experimental and Computational Studies on the C-H Amination Mechanism of Tetrahydrocarbazoles via Hydroperoxides
N. Gulzar, K. M. Jones, H. Konnerth, M. Breugst,* M. Klussmann,* Chem. Eur. J. 2015, 21, 3367–3376.
DOI: 10.1002/chem.201405376
N. Gulzar, K. M. Jones, H. Konnerth, M. Breugst,* M. Klussmann,* Chem. Eur. J. 2015, 21, 3367–3376.
DOI: 10.1002/chem.201405376

2014
14. Computational Analysis of Cyclophane-Based Bisthiourea-Catalyzed Henry Reactions
M. Breugst,* K. N. Houk* J. Org. Chem. 2014, 79, 6302–6309.
DOI: 10.1021/jo501227m
M. Breugst,* K. N. Houk* J. Org. Chem. 2014, 79, 6302–6309.
DOI: 10.1021/jo501227m

13. Redox-Neutral α-Sulfenylation of Secondary Amines: Ring-Fused N,S-Acetals
C. L. Jarvis, M. T. Richers, M. Breugst, K. N. Houk, D. Seidel, Org. Lett. 2014, 16, 3556–3559.
DOI: 10.1021/ol501509b
C. L. Jarvis, M. T. Richers, M. Breugst, K. N. Houk, D. Seidel, Org. Lett. 2014, 16, 3556–3559.
DOI: 10.1021/ol501509b

12. Redox-Neutral α-Oxygenation of Amines: Reaction Development and Elucidation of the Mechanism
M. T. Richers, M. Breugst, A. Y. Platonova, A. Ullrich, A. Dieckmann, K. N. Houk, D. Seidel, J. Am. Chem. Soc. 2014, 136, 6123–6135.
DOI: 10.1021/ja501988b
M. T. Richers, M. Breugst, A. Y. Platonova, A. Ullrich, A. Dieckmann, K. N. Houk, D. Seidel, J. Am. Chem. Soc. 2014, 136, 6123–6135.
DOI: 10.1021/ja501988b

11. δ-Deuterium Isotope Effects as Probes for Transition State Structures of Isoprenoid Substrates
S.-r. Choi, M. Breugst, K. N. Houk, C. D. Poulter, J. Org. Chem. 2014, 79, 3572–3580.
DOI: 10.1021/jo500394u
S.-r. Choi, M. Breugst, K. N. Houk, C. D. Poulter, J. Org. Chem. 2014, 79, 3572–3580.
DOI: 10.1021/jo500394u

Earlier Publications
10. Synergistic Effects Between Lewis and Brønsted Acids – Application to the Prins Cyclization
M. Breugst,* R. Grée, K. N. Houk,* J. Org. Chem. 2013, 78, 9892–9897.
DOI: 10.1021/jo401628e
M. Breugst,* R. Grée, K. N. Houk,* J. Org. Chem. 2013, 78, 9892–9897.
DOI: 10.1021/jo401628e

09. Theoretical Exploration of the Mechanism of Riboflavin Formation from 6,7-Dimethyl-8-ribityl-lumazine: Hydride Transfer, Hydrogen Atom Transfer, Nucleophilic Addition, or Nucleophilic Catalysis?
M. Breugst, A. Eschenmoser, K. N. Houk, J. Am. Chem. Soc. 2013, 135, 6658–6668.
DOI: 10.1021/ja402099f
M. Breugst, A. Eschenmoser, K. N. Houk, J. Am. Chem. Soc. 2013, 135, 6658–6668.
DOI: 10.1021/ja402099f

08. Zwitterions and Unobserved Intermediates in Organocatalytic Diels-Alder Reactions of Linear and Cross-Conjugated Trienamines
A. Dieckmann, M. Breugst, K. N. Houk, J. Am. Chem. Soc. 2013, 135, 3237–3242.
DOI: 10.1021/ja312043g
Highlighted in Comp. Chem. Highlights, 2013, 2013.04.zwitterions.
A. Dieckmann, M. Breugst, K. N. Houk, J. Am. Chem. Soc. 2013, 135, 3237–3242.
DOI: 10.1021/ja312043g
Highlighted in Comp. Chem. Highlights, 2013, 2013.04.zwitterions.

07. Nucleophilic Reactivities of the Anions of Nucleobases and Their Subunits
M. Breugst, F. Corral Bautista, H. Mayr, Chem. Eur. J. 2012, 18, 127–137.
DOI: 10.1002/chem.201102411
Selected as a VIP Paper.
M. Breugst, F. Corral Bautista, H. Mayr, Chem. Eur. J. 2012, 18, 127–137.
DOI: 10.1002/chem.201102411
Selected as a VIP Paper.

06. N-Heterocyclic Carbenes: Organocatalysts with Moderate Nucleophilicity but Extraordinarily High Lewis Basicity
B. Maji, M. Breugst, H. Mayr, Angew. Chem. Int. Ed. 2011, 50, 6915–6919; Angew. Chem. 2011, 123, 7047–7052.
DOI: 10.1002/anie.201102435
B. Maji, M. Breugst, H. Mayr, Angew. Chem. Int. Ed. 2011, 50, 6915–6919; Angew. Chem. 2011, 123, 7047–7052.
DOI: 10.1002/anie.201102435
05. Farewell to the HSAB Treatment of Ambident Reactivity
H. Mayr, M. Breugst, A. R. Ofial, Angew. Chem. Int. Ed. 2011, 50, 6470–6505; Angew. Chem. 2011, 123, 6598–6634.
DOI: 10.1002/anie.201007100
H. Mayr, M. Breugst, A. R. Ofial, Angew. Chem. Int. Ed. 2011, 50, 6470–6505; Angew. Chem. 2011, 123, 6598–6634.
DOI: 10.1002/anie.201007100

04. Ambident Reactivities of Pyridone Anions
M. Breugst, H. Mayr, J. Am. Chem. Soc. 2010, 132, 15380–15389.
DOI: 10.1021/ja106962u
M. Breugst, H. Mayr, J. Am. Chem. Soc. 2010, 132, 15380–15389.
DOI: 10.1021/ja106962u

03. Nucleophilic Reactivities of Imide and Amide Anions
M. Breugst, T. Tokuyasu, H. Mayr, J. Org. Chem. 2010, 75, 5050–5258.
DOI: 10.1021/jo1009883
M. Breugst, T. Tokuyasu, H. Mayr, J. Org. Chem. 2010, 75, 5050–5258.
DOI: 10.1021/jo1009883

02. Marcus-Analysis of Ambident Reactivity
M. Breugst, H. Zipse, J. P. Guthrie, H. Mayr, Angew. Chem. Int. Ed. 2010, 49, 5165–5169; Angew. Chem. 2010, 122, 5291–5295. DOI: 10.1002/anie.201001574
M. Breugst, H. Zipse, J. P. Guthrie, H. Mayr, Angew. Chem. Int. Ed. 2010, 49, 5165–5169; Angew. Chem. 2010, 122, 5291–5295. DOI: 10.1002/anie.201001574

01. Towards a General Scale of Nucleophilicity?
T. B. Phan, M. Breugst, H. Mayr, Angew. Chem. Int. Ed. 2006, 45, 3869–3874; Angew. Chem. 2006, 118, 3954–3959.
DOI: 10.1002/anie.200600542
Selected as a VIP Paper.
T. B. Phan, M. Breugst, H. Mayr, Angew. Chem. Int. Ed. 2006, 45, 3869–3874; Angew. Chem. 2006, 118, 3954–3959.
DOI: 10.1002/anie.200600542
Selected as a VIP Paper.


